 The electrons revolve around the nucleus in circular paths.Nearly all the mass of an atom resides in the nucleus. There is a positively charged centre in an atom called the nucleus.On the basis of his experiment, Rutherford put forward the nuclear model of an atom, which had the following features: In this experiment, fast moving alpha (α)-particles were made to fall on a thin gold foil. Rutherford designed an experiment for this. Ernest Rutherford was interested in knowing how the electrons are arranged within an atom. He is famous for his work on radioactivity and the discovery of the nucleus of an atom with the gold foil experiment. Rutherford (1871-1937) was known as the ‘Father’ of nuclear physics. The red part of the watermelon as positively charged.Watermelon seeds as negatively charged particles.Thomson atomic model is compared to watermelon. Postulate 2: An atom as a whole is electrically neutral because the negative and positive charges are equal in magnitude Postulate 1: An atom consists of a positively charged sphere with electrons embedded in it. (5) Thomson was the first one to propose a model for the structure of an atom: But the discovery of two fundamental particles (electrons and protons) inside the atom, led to the failure of this aspect of Dalton’s atomic theory. (4) Dalton’s atomic theory suggested that the atom was indivisible and indestructible. Thomson’s model for the structure of an atom? (4) “Atom is indivisible and indestructible” why this aspect of Dalton’s atomic theory leds to the failure? (3) Who was the first to propose atomic theory? (2) According to Dalton’s Atomic Theory, matter consists of indivisible _ Statement 4 – Protons are positively charged particle. Thomson was the first one to propose a model for the structure of an atom. Statement 2 – Electrons and protons are present inside the atom. Statement 1 – Dalton’s atomic theory suggested that the atom was indivisible and indestructible. So, the atom as a whole is electrically neutral. The negative and positive charges are equal in magnitude. Thomson proposed that: An atom consists of a positively charged sphere and the electrons are embedded in it. We can also think of a watermelon, the positive charge in the atom is spread all over like the red edible part of the watermelon, while the electrons are studded in the positively charged sphere, like the seeds in the watermelon. The electrons, in a sphere of positive charge. Thomson proposed the model of an atom to be similar to that of a Christmas pudding. Thomson (1856- 1940) was a British physicist, He was awarded the Nobel Prize in Physics for his work on the discovery of electrons. 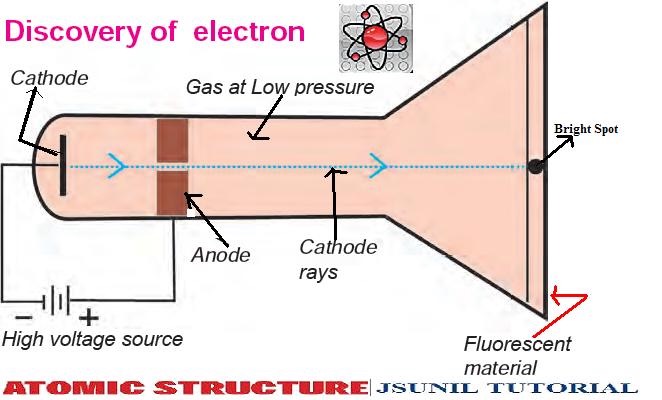
For explaining this, many scientists proposed various atomic models. It was then considered necessary to know how electrons and protons are arranged within an atom. CBSE Case Study Questions Class 9 Science – Structure of the Atomĭalton’s atomic theory suggested that the atom was indivisible and indestructible. 
There will various types of marks will given 1 marks, 2 marks, 3 marks or 4 marks. In where some Questions will made on that respective Case Based Study. Here we have arranged some Important Case Base Questions for students who are searching for Paragraph Based Questions Structure of the Atom.Īt Case Study Questions there will given a Paragraph. Important Case Study Questions for Class 9 Exam. Chemistry quiz | Chemistry Questions | Chemicals question answer Case Study Questions Class 9 Science Chapter 4 Structure of the AtomĬBSE Class 9 Case Study Questions Science Structure of the Atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
